Batteries are stores of energy that become electricity when we release it. This amount of energy is tiny when we compare it to the grid. However unlike power stations and grids, batteries are portable. In fact batteries and wearable solar panels are probably the only powerhouses we could take with us anywhere we go. These basic battery facts could not exist without electro chemistry.
Basic Battery Facts & What Goes On Inside
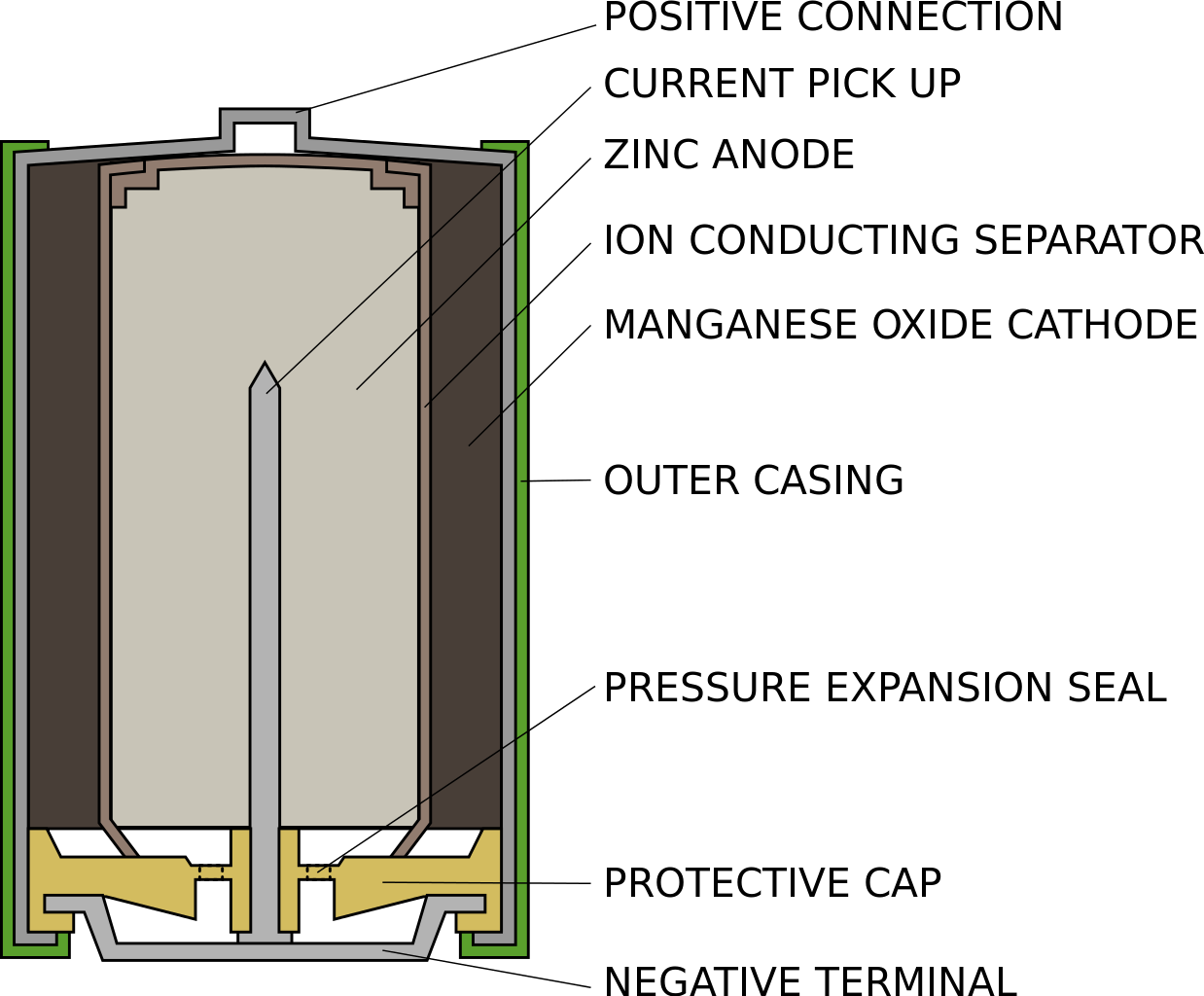
The batteries we purchase in the stores all have four basic parts inside the case. First, there are two electrodes. The cathode electrode connects to the positive terminal. While the anode one joins to the other one.
A ‘gatekeeper’ electrolyte sits between them containing tiny electrically charged ions. Finally, a separator prevents the electrodes touching. When we connect a device to a battery, we create a circuit that releases energy through it. These are the basic battery facts.
Single Cycle and Rechargeable Batteries

Nowadays most batteries are rechargeable, along the same lines as the lead-acid batteries we sell. This means we can put the energy back into them after we discharge them. We call a charge followed by a discharge a ‘cycle’. This is not the same as recycling the materials in a dead battery
Each cycle reduces the ability of a battery to reach full charge again. A battery is technically ‘dead’ when it can no longer hold enough power to operate a matching device. Hence, when we talk about cycle life under battery basics we refer to the number of complete cycles it should manage.
Batteries and the Future of Grid Storage
Grid storage lithium and flow batteries are becoming larger and more expensive. Although it is also true their cost per gigawatt-hour is falling due to economies of scale. The future of alternative energy hangs on increasing their cycle life. And that in turn depends on the basic battery facts we describe in this post.
Related
How Single Cycle Batteries Go Flat
How to Charge a Lead-Acid Battery Safely
Preview Image: Mercury Battery




