Lithium has made the headlines lately in ways that are not of its own making. Scientists have been pairing it with other elements in a bid to make the most efficient set of electrodes. From time to time, these have overheated putting Samsung Note 7’s and Boeing Dreamliners on the tip of everybody’s tongue. So what is this strange substance we call Lithium Li?
Lithium Li is One of Nature’s Building Blocks

Lithium has an atomic number three. This means there are only three protons in its nucleus making it an extremely simple structure. Despite this, it is the lightest metal that we know of, and the least-dense solid element.
Like other alkaline metals, Lithium Li also has a bad temper chemically speaking. Being an alkaline metal makes it highly reactive and flammable. The safest way to store it is in mineral oil in which it floats. A cut surface initially displays a metallic sheen. This quickly fades to dull slivery grey, and then black tarnish. Here’s a Wikipedia link if you would like to know more about its chemical structure.
Lithium is Not Just for Lithium-Ion Batteries
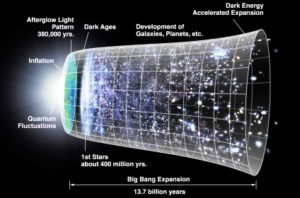
Big Bang theory holds that Lithium Li was one of three elements created at the foundation of the cosmos. Although this is only a theory, the silvery metal does appear widespread across the universe.
It occurs extensively on earth and in the ocean. We do not find it in its elemental form, because its bad temper could cause it to self-destruct.
The military use the reactive nature of lithium to its advantage in rocket propellants and torpedo power. A version of Lithium Li was the fusion fuel for early hydrogen bombs. Some nuclear power stations use a derivative to create controlled nuclear reactions. On a more positive note, Lithium salts help alleviate bipolar and schizo disorders, and also cyclical depressions.
Related
Was Samsung’s Lithium-Ion Technology the Cause
Scientists Develop Cross-Linked Electrolytes … Batteries Safer




