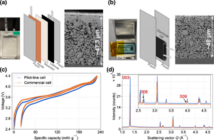
While we charge a lithium battery, the positively charged atoms we call ‘ions’ transfer from the lithium to the carbon electrode. When we consume the energy this creates, they flow the other way. Most batteries work on similar principles although the design and materials vary. Much research focuses on improving this process including using potassium ions.
Battery charging efficiency depends on the speed with which the lithium ions return to the lithium electrode for recycling. Moreover, they do not all find their way back to the electrode, resulting in progressively lower battery density and faster discharge. Scientists are working hard at resolving these issues and because of this we are making progress.
Good Progress with Potassium Ions in Bath

The City of Bath in Somerset, England is famous for its curative waters, ancient ruins, and high property prices.
In the midst of this enviable splendor, it has a leading university that is internationally renowned for teaching and research excellence.
It is currently creating an institute for advanced automotive propulsion systems. This line of thinking may be what took it down the route of tinkering with lithium batteries.
We do not know whether their discovery was the result of research, or inspiration. Science certainly is a mixture of the two. What we do know is they discovered that inserting positive potassium ions inside rechargeable lithium batteries improves their electrical conductivity, and hence recharging speed.
The Road Ahead for the Potassium Breakthrough

Lead researcher Prof Saiful Islam is a man with a mission. “Developing new materials holds the key to lighter, cheaper and safer batteries,” he says. “And this includes for electric vehicles which will help cut carbon emissions.”
He plans to continue collaborating with colleagues from the University of Illinois in Chicago. His next step is to find out why inserting charged potassium ions makes such a huge difference. We wonder what inspired him to choose potassium ions in the first place.
Related
Longer Lithium Battery Life Upon Us
Principles of Extending Lithium Battery Life
Preview Image: Potassium in Feldspar