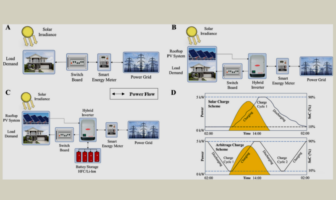
 Battery Technical
Battery Technical
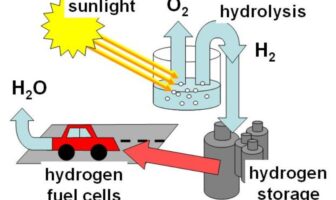
Hydrogen Solar Storage Versus Lithium-Ion
Metal-hydride batteries use hydrogen-storage-alloys as their negative electrodes. This arrangement delivers relatively higher density, or…
 Battery Technical
Battery Technical
Metal-hydride batteries use hydrogen-storage-alloys as their negative electrodes. This arrangement delivers relatively higher density, or…
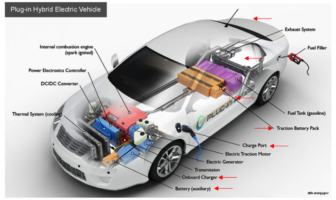 Battery Basics
Battery Basics
If you’re shopping for prices for a new electric auto battery on the internet, the…
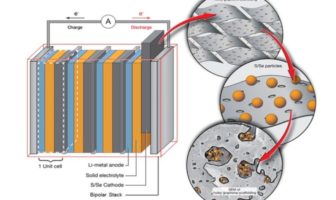 Battery Technical
Battery Technical
There’s much talk in media about solid-state electrolyte. We review why solid state could side-step…
 Battery Basics
Battery Basics
The European Joint Review Committee (JRC) has oversight over individual EU member state reports. It…
 Battery Technical
Battery Technical
China’s BYD electric vehicle maker is in a tight race with Tesla to have higher…
 Battery Technical
Battery Technical
Chair of India’s Science, Technology, and Innovation Advisory Council, Ajay Sood, is a man with…
 Battery Technical
Battery Technical
There are various types of sodium-ion batteries on the market, which use their ions as…
 Featured
Featured
Climate change could severely affect the future of remote, far-flung islands, if warming continues. Many…
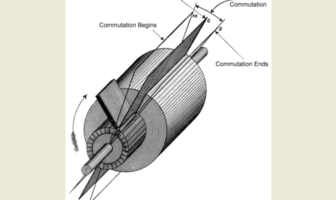 Battery Basics
Battery Basics
Dynamos and alternators produce different currents, namely direct and alternating electricity. In other words D…
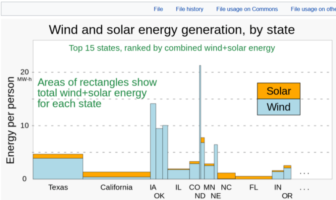 Featured
Featured
Which is the best renewable source for my home or business, our readers often ask.…
